-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
The Ten-Year Choice: Why Younger Heart Patients Are Rethinking TAVR and Returning to the Operating Table
The Promise and the Problem
When transcatheter aortic valve replacement emerged from the catheterization laboratory into mainstream clinical practice in the early 2010s, it carried the weight of a genuine revolution. For the first time, patients with severe, symptomatic aortic stenosis who were deemed too frail, too elderly, or too comorbid for the trauma of open-heart surgery could receive a functional valve replacement through a catheter threaded from the groin — without a sternal incision, without cardiopulmonary bypass, without the weeks of recovery that traditional surgical valve replacement demands. The early outcomes were transformative. Survival rates in inoperable and high-risk surgical patients improved dramatically, and the procedure's technical refinement over the following decade extended its application progressively toward lower-risk patient populations.
That extension has now reached a point where it demands a more careful conversation than the straightforward calculus of mortality and morbidity that governed the early TAVR era. As the procedural eligibility criteria have expanded to include intermediate-risk and low-risk patients — and as some centers have begun performing TAVR in patients in their fifties — a different question has moved to the foreground: not whether the patient can survive the procedure, but whether the valve they receive will survive the patient. The answer, increasingly and uncomfortably, is that for many younger patients, it will not.
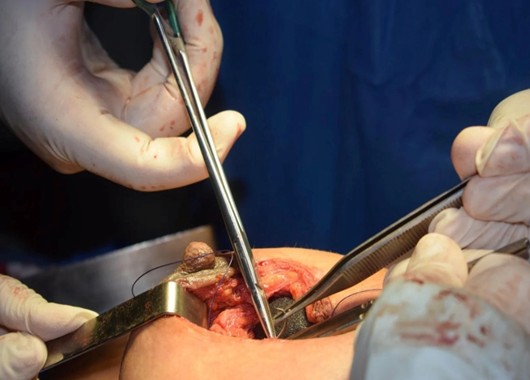
This article examines the emerging clinical and evidence-based tension between the procedural convenience of TAVR and the long-term durability requirements of patients whose life expectancy may span four or five decades beyond their valve implant date. It argues that the open surgical field depicted in Figure 1 — imposing, technical, and demanding — is not an anachronism in the age of transcatheter intervention but a deliberate and evidence-informed choice for a specific and growing patient population: the younger patient with aortic stenosis who needs not just a valve, but a valve that will last.
That extension has now reached a point where it demands a more careful conversation than the straightforward calculus of mortality and morbidity that governed the early TAVR era. As the procedural eligibility criteria have expanded to include intermediate-risk and low-risk patients — and as some centers have begun performing TAVR in patients in their fifties — a different question has moved to the foreground: not whether the patient can survive the procedure, but whether the valve they receive will survive the patient. The answer, increasingly and uncomfortably, is that for many younger patients, it will not.
This article examines the emerging clinical and evidence-based tension between the procedural convenience of TAVR and the long-term durability requirements of patients whose life expectancy may span four or five decades beyond their valve implant date. It argues that the open surgical field depicted in Figure 1 — imposing, technical, and demanding — is not an anachronism in the age of transcatheter intervention but a deliberate and evidence-informed choice for a specific and growing patient population: the younger patient with aortic stenosis who needs not just a valve, but a valve that will last.
Aortic Stenosis in Younger Patients: An Evolving Epidemiology
Aortic stenosis has historically been framed as a disease of the elderly — the consequence of decades of calcium deposition on the leaflets of a tricuspid aortic valve, producing the progressive obstruction of left ventricular outflow that leads to the classic symptomatic triad of angina, syncope, and heart failure. This framing is epidemiologically accurate for the majority of patients, in whom degenerative calcific aortic stenosis manifests in the seventh to ninth decades of life. It is, however, an incomplete picture of the contemporary landscape, particularly in younger populations where bicuspid aortic valve disease — the most common congenital cardiac anomaly, present in approximately one to two percent of the population — accelerates the degenerative process and produces severe aortic stenosis requiring intervention in the fifth and sixth decades.
Bicuspid aortic valve disease deserves specific attention in this context for reasons that go beyond its younger age of presentation. The abnormal valve architecture of a bicuspid aortic valve — with its fused leaflets, asymmetric commissures, and high-velocity eccentric jet during systole — produces a valve morphology that is technically challenging for transcatheter prosthesis deployment, and the associated aortopathy — dilation of the ascending aorta that accompanies bicuspid aortic valve disease in a substantial proportion of patients — may require concomitant surgical management that cannot be addressed through a catheter-based approach. A young patient with bicuspid aortic stenosis and an ascending aorta measuring 4.5 centimeters presents a composite surgical problem for which open surgical repair offers the only complete solution.
Beyond bicuspid disease, rheumatic aortic stenosis — increasingly rare in high-income countries but still prevalent in parts of South and Southeast Asia, sub-Saharan Africa, and Latin America — produces valve disease in young adults that requires intervention in the third and fourth decades of life, placing the durability question in an even starker demographic context. A thirty-five-year-old patient with rheumatic aortic stenosis who receives a valve replacement can reasonably expect to live another fifty years — an expectation that transforms the question of valve durability from a ten-year concern into a multi-decade planning problem
Bicuspid aortic valve disease deserves specific attention in this context for reasons that go beyond its younger age of presentation. The abnormal valve architecture of a bicuspid aortic valve — with its fused leaflets, asymmetric commissures, and high-velocity eccentric jet during systole — produces a valve morphology that is technically challenging for transcatheter prosthesis deployment, and the associated aortopathy — dilation of the ascending aorta that accompanies bicuspid aortic valve disease in a substantial proportion of patients — may require concomitant surgical management that cannot be addressed through a catheter-based approach. A young patient with bicuspid aortic stenosis and an ascending aorta measuring 4.5 centimeters presents a composite surgical problem for which open surgical repair offers the only complete solution.
Beyond bicuspid disease, rheumatic aortic stenosis — increasingly rare in high-income countries but still prevalent in parts of South and Southeast Asia, sub-Saharan Africa, and Latin America — produces valve disease in young adults that requires intervention in the third and fourth decades of life, placing the durability question in an even starker demographic context. A thirty-five-year-old patient with rheumatic aortic stenosis who receives a valve replacement can reasonably expect to live another fifty years — an expectation that transforms the question of valve durability from a ten-year concern into a multi-decade planning problem
The TAVR Revolution: What It Achieved and Where It Excels
Before examining the limitations of TAVR in younger patients, it is important to be precise about what transcatheter aortic valve replacement has achieved and where it continues to represent the unambiguously superior treatment strategy. In patients over seventy-five with severe symptomatic aortic stenosis and high or prohibitive surgical risk — defined by frailty, severe chronic obstructive pulmonary disease, prior radiation to the chest, porcelain aorta, or the constellation of organ dysfunction that places open surgery beyond safe tolerance — TAVR has demonstrably reduced mortality, reduced procedural morbidity, shortened hospital stay, and preserved quality of life in the months and years following intervention.
The PARTNER and CoreValve trial programs, together with their subsequent iterative extensions into intermediate and low surgical risk populations, established the evidence base that has driven TAVR from an experimental rescue procedure to the dominant modality for aortic valve replacement in Europe and North America. In elderly patients, the ten-year durability horizon is clinically acceptable: an eighty-year-old patient who receives a TAVR prosthesis at high surgical risk and achieves good hemodynamic outcomes for the eight to twelve years that the valve is expected to function reliably has received a treatment that is well matched to their life expectancy and their tolerance for procedural risk.
The procedural advantages of TAVR in this population are genuine and not to be dismissed. The elimination of cardiopulmonary bypass removes the systemic inflammatory response, the coagulation cascade activation, and the neurological injury risk associated with the heart-lung machine. The avoidance of sternotomy eliminates the sternal wound complications, the pain, and the six-week activity restriction of open surgery. The twenty-four to forty-eight-hour hospital stay of modern transfemoral TAVR versus the five to seven days of surgical aortic valve replacement represents a meaningful difference in healthcare resource utilization and patient experience. For the right patient — elderly, frail, high surgical risk — these advantages are decisive.
The PARTNER and CoreValve trial programs, together with their subsequent iterative extensions into intermediate and low surgical risk populations, established the evidence base that has driven TAVR from an experimental rescue procedure to the dominant modality for aortic valve replacement in Europe and North America. In elderly patients, the ten-year durability horizon is clinically acceptable: an eighty-year-old patient who receives a TAVR prosthesis at high surgical risk and achieves good hemodynamic outcomes for the eight to twelve years that the valve is expected to function reliably has received a treatment that is well matched to their life expectancy and their tolerance for procedural risk.
The procedural advantages of TAVR in this population are genuine and not to be dismissed. The elimination of cardiopulmonary bypass removes the systemic inflammatory response, the coagulation cascade activation, and the neurological injury risk associated with the heart-lung machine. The avoidance of sternotomy eliminates the sternal wound complications, the pain, and the six-week activity restriction of open surgery. The twenty-four to forty-eight-hour hospital stay of modern transfemoral TAVR versus the five to seven days of surgical aortic valve replacement represents a meaningful difference in healthcare resource utilization and patient experience. For the right patient — elderly, frail, high surgical risk — these advantages are decisive.
The Durability Problem: What Happens When the Valve Wears Out
Structural Valve Deterioration: The Biology of Bioprosthetic Failure
All currently available TAVR prostheses use biological tissue — pericardium from bovine or porcine sources, treated with glutaraldehyde fixation to reduce antigenicity and sterilize the material — as the leaflet material. This biological fixation process, while essential for commercial production and implantation, has a critical long-term consequence: it eliminates the viability of the leaflet tissue. Fixed bioprosthetic leaflets are not living tissue capable of remodeling and self-repair; they are biological scaffolds that degrade progressively under the mechanical stress of three billion cardiac cycles per decade, the calcium deposition driven by chronic leaflet microtrauma and phospholipid exposure, and the oxidative injury produced by the chronically inflamed environment of the implanted valve.
Structural valve deterioration — the failure mode characterized by calcific leaflet thickening, reduced leaflet mobility, increased transvalvular gradient, or progressive leaflet regurgitation — is the dominant long-term failure mode of bioprosthetic valves. Its incidence is age-dependent in a biologically important direction: younger patients experience faster bioprosthetic deterioration than older ones, almost certainly because of more vigorous calcium metabolism, higher cardiac output demands producing greater mechanical stress per cycle, and potentially different inflammatory responses to the foreign biological material. Studies of surgically implanted bioprosthetic valves — for which longer follow-up data exist than for TAVR prostheses — demonstrate freedom from structural valve deterioration of approximately ninety percent at ten years, seventy percent at fifteen years, and fifty percent or less at twenty years, with significantly worse outcomes in patients implanted at younger ages.
Longer-term durability data for TAVR prostheses specifically are beginning to emerge from the cohorts enrolled in the pivotal trials, and they are sobering. Five-year data from PARTNER and CoreValve trials demonstrate structural valve deterioration rates of ten to fifteen percent, and early ten-year data from centers with the longest TAVR follow-up suggest continued attrition beyond this timeframe. The relatively recent introduction of TAVR to younger, lower-risk populations means that the full magnitude of the durability problem in this demographic will not be known from trial data for another decade — but the biological principles that govern bioprosthetic durability leave little reason for optimism about outcomes in patients implanted at fifty or fifty-five.
All currently available TAVR prostheses use biological tissue — pericardium from bovine or porcine sources, treated with glutaraldehyde fixation to reduce antigenicity and sterilize the material — as the leaflet material. This biological fixation process, while essential for commercial production and implantation, has a critical long-term consequence: it eliminates the viability of the leaflet tissue. Fixed bioprosthetic leaflets are not living tissue capable of remodeling and self-repair; they are biological scaffolds that degrade progressively under the mechanical stress of three billion cardiac cycles per decade, the calcium deposition driven by chronic leaflet microtrauma and phospholipid exposure, and the oxidative injury produced by the chronically inflamed environment of the implanted valve.
Structural valve deterioration — the failure mode characterized by calcific leaflet thickening, reduced leaflet mobility, increased transvalvular gradient, or progressive leaflet regurgitation — is the dominant long-term failure mode of bioprosthetic valves. Its incidence is age-dependent in a biologically important direction: younger patients experience faster bioprosthetic deterioration than older ones, almost certainly because of more vigorous calcium metabolism, higher cardiac output demands producing greater mechanical stress per cycle, and potentially different inflammatory responses to the foreign biological material. Studies of surgically implanted bioprosthetic valves — for which longer follow-up data exist than for TAVR prostheses — demonstrate freedom from structural valve deterioration of approximately ninety percent at ten years, seventy percent at fifteen years, and fifty percent or less at twenty years, with significantly worse outcomes in patients implanted at younger ages.
Longer-term durability data for TAVR prostheses specifically are beginning to emerge from the cohorts enrolled in the pivotal trials, and they are sobering. Five-year data from PARTNER and CoreValve trials demonstrate structural valve deterioration rates of ten to fifteen percent, and early ten-year data from centers with the longest TAVR follow-up suggest continued attrition beyond this timeframe. The relatively recent introduction of TAVR to younger, lower-risk populations means that the full magnitude of the durability problem in this demographic will not be known from trial data for another decade — but the biological principles that govern bioprosthetic durability leave little reason for optimism about outcomes in patients implanted at fifty or fifty-five.
Valve-in-Valve TAVR: The Redo Procedure and Its Limitations
When a TAVR prosthesis fails, the primary reintervention strategy is valve-in-valve TAVR — the deployment of a new transcatheter prosthesis within the failing original one. This technique is technically feasible and has become standard practice at experienced centers, avoiding the need for reoperation in patients who received their initial TAVR because they were deemed high surgical risk. In the elderly, high-risk patient for whom TAVR was originally designed, valve-in-valve is an appropriate and effective management strategy for structural valve deterioration.
In younger patients, however, valve-in-valve TAVR carries limitations that significantly diminish its attractiveness as a planned reintervention strategy. The most concerning is coronary obstruction: when a new transcatheter prosthesis is deployed within a degenerated TAVR valve, the displaced leaflet material of the original prosthesis can occlude the coronary ostia — the origins of the coronary arteries — producing catastrophic or fatal coronary ischemia. The risk of coronary obstruction in valve-in-valve procedures is determined primarily by anatomical factors including the distance between the valve annulus and the coronary ostia (the virtual transcatheter heart valve to coronary artery distance, or VTC distance) and the size and design of the original prosthesis. A significant proportion of patients — estimated at twenty to thirty percent, depending on valve selection and anatomy — have anatomy that is unfavorable for safe valve-in-valve TAVR, potentially condemning them to higher-risk redo open surgery at a time when their overall cardiovascular status may be substantially worse than at the time of their original intervention.
Beyond coronary obstruction, valve-in-valve procedures are inherently associated with higher residual gradients than index TAVR procedures, because the effective orifice area of the new prosthesis is constrained by the internal diameter of the degenerated original. For a young, physically active patient with a normal or high cardiac output requirement, a residual mean gradient of fifteen to twenty millimeters of mercury — not uncommon after valve-in-valve TAVR in small annular sizes — may be hemodynamically acceptable at rest but produce symptomatic limitation during exercise that compromises quality of life and physical capacity.
In younger patients, however, valve-in-valve TAVR carries limitations that significantly diminish its attractiveness as a planned reintervention strategy. The most concerning is coronary obstruction: when a new transcatheter prosthesis is deployed within a degenerated TAVR valve, the displaced leaflet material of the original prosthesis can occlude the coronary ostia — the origins of the coronary arteries — producing catastrophic or fatal coronary ischemia. The risk of coronary obstruction in valve-in-valve procedures is determined primarily by anatomical factors including the distance between the valve annulus and the coronary ostia (the virtual transcatheter heart valve to coronary artery distance, or VTC distance) and the size and design of the original prosthesis. A significant proportion of patients — estimated at twenty to thirty percent, depending on valve selection and anatomy — have anatomy that is unfavorable for safe valve-in-valve TAVR, potentially condemning them to higher-risk redo open surgery at a time when their overall cardiovascular status may be substantially worse than at the time of their original intervention.
Beyond coronary obstruction, valve-in-valve procedures are inherently associated with higher residual gradients than index TAVR procedures, because the effective orifice area of the new prosthesis is constrained by the internal diameter of the degenerated original. For a young, physically active patient with a normal or high cardiac output requirement, a residual mean gradient of fifteen to twenty millimeters of mercury — not uncommon after valve-in-valve TAVR in small annular sizes — may be hemodynamically acceptable at rest but produce symptomatic limitation during exercise that compromises quality of life and physical capacity.
The Surgical Alternative: Mechanical Valves and the Anticoagulation Trade-Off
The traditional surgical alternative to bioprosthetic valve replacement in young patients is the mechanical prosthesis — a valve constructed from pyrolytic carbon leaflets with a metal housing, capable of functioning for decades beyond the lifespan of any currently available biological tissue valve. Modern bileaflet mechanical valves — the St. Jude Medical Regent, the On-X, the CarboMedics — have demonstrated freedom from structural valve deterioration exceeding ninety-five percent at twenty years, and the oldest implanted mechanical valves have now been functioning continuously for more than four decades in some patients. For a forty-five-year-old patient with severe aortic stenosis who expects to live another four decades, the mechanical valve's durability profile is fundamentally different from that of any bioprosthetic alternative.
The cost of this durability is mandatory lifelong anticoagulation with a vitamin K antagonist — warfarin or acenocoumarol — to prevent thromboembolism on the non-biological valve surfaces. The burden of warfarin anticoagulation is real and not to be minimized: regular INR monitoring, dietary interactions, drug-drug interactions, the two to three percent per year risk of serious bleeding complications, and the absolute contraindication to pregnancy that warfarin carries in the first trimester due to warfarin embryopathy. For the right patient — one who is organized, medically engaged, not planning a pregnancy, and who understands the trade-off between anticoagulation burden and the near-certainty of valve durability — this is an entirely rational choice.
A significant recent development in mechanical valve management has been the PROACT Xa trial of the On-X mechanical aortic valve, which demonstrated that patients implanted with this specific valve could be safely managed with warfarin targeting a lower INR range (1.5 to 2.0 rather than the conventional 2.0 to 3.0) combined with low-dose aspirin, reducing the annual bleeding risk substantially compared to conventional anticoagulation intensity without an increase in thromboembolic events. This finding, if confirmed in broader follow-up and extended to other valve designs, has the potential to significantly improve the risk-benefit calculus of mechanical valve selection in young patients by reducing the most clinically burdensome aspect of the anticoagulation commitment.
The Ross procedure — an alternative surgical strategy in which the patient's own pulmonary valve is harvested and reimplanted in the aortic position, with a biological conduit placed in the pulmonary position — deserves mention as a particularly attractive option in younger patients who wish to avoid both the anticoagulation burden of a mechanical prosthesis and the durability limitations of a biological one. By replacing the aortic valve with the patient's own living pulmonary valve tissue, the Ross procedure provides a valve that can grow with the patient (relevant in pediatric cases), remodel in response to the hemodynamic environment of the systemic circulation, and remain free from the degeneration that affects fixed biological tissue. Long-term outcomes from experienced centers demonstrate Ross procedure durability superior to that of bioprosthetic valves in young patients, though the complexity of the procedure — it involves two valve positions rather than one — means that results are highly center-volume-dependent and that widespread application requires experienced surgical teams.
The cost of this durability is mandatory lifelong anticoagulation with a vitamin K antagonist — warfarin or acenocoumarol — to prevent thromboembolism on the non-biological valve surfaces. The burden of warfarin anticoagulation is real and not to be minimized: regular INR monitoring, dietary interactions, drug-drug interactions, the two to three percent per year risk of serious bleeding complications, and the absolute contraindication to pregnancy that warfarin carries in the first trimester due to warfarin embryopathy. For the right patient — one who is organized, medically engaged, not planning a pregnancy, and who understands the trade-off between anticoagulation burden and the near-certainty of valve durability — this is an entirely rational choice.
A significant recent development in mechanical valve management has been the PROACT Xa trial of the On-X mechanical aortic valve, which demonstrated that patients implanted with this specific valve could be safely managed with warfarin targeting a lower INR range (1.5 to 2.0 rather than the conventional 2.0 to 3.0) combined with low-dose aspirin, reducing the annual bleeding risk substantially compared to conventional anticoagulation intensity without an increase in thromboembolic events. This finding, if confirmed in broader follow-up and extended to other valve designs, has the potential to significantly improve the risk-benefit calculus of mechanical valve selection in young patients by reducing the most clinically burdensome aspect of the anticoagulation commitment.
The Ross procedure — an alternative surgical strategy in which the patient's own pulmonary valve is harvested and reimplanted in the aortic position, with a biological conduit placed in the pulmonary position — deserves mention as a particularly attractive option in younger patients who wish to avoid both the anticoagulation burden of a mechanical prosthesis and the durability limitations of a biological one. By replacing the aortic valve with the patient's own living pulmonary valve tissue, the Ross procedure provides a valve that can grow with the patient (relevant in pediatric cases), remodel in response to the hemodynamic environment of the systemic circulation, and remain free from the degeneration that affects fixed biological tissue. Long-term outcomes from experienced centers demonstrate Ross procedure durability superior to that of bioprosthetic valves in young patients, though the complexity of the procedure — it involves two valve positions rather than one — means that results are highly center-volume-dependent and that widespread application requires experienced surgical teams.
The Low-Risk Surgical Evidence: Reclaiming Open Surgery’s Position
The expansion of TAVR to low surgical risk patients was driven in part by trial evidence from PARTNER 3 and Evolut Low Risk, which demonstrated non-inferiority of TAVR compared to surgical aortic valve replacement for thirty-day and two-year composite endpoints of mortality, stroke, and rehospitalization in low-risk patients. These results were interpreted by many in the cardiology community as evidence that TAVR should be offered to all low-risk patients, including younger ones. A more careful reading of the data, however, reveals that the follow-up period of these trials was insufficient to capture the durability divergence between transcatheter and surgical prostheses that is expected to manifest at five to ten years and beyond.
Surgical aortic valve replacement in low-risk patients, particularly at experienced centers with high surgical volumes, carries thirty-day mortality rates of well under one percent and stroke rates of one to two percent — outcomes that are equivalent to or approaching those of TAVR in the same risk category, and that are achieved with the implantation of a prosthesis whose durability profile is substantially better defined and longer-established than that of any currently available TAVR device. The modern era of minimally invasive surgical aortic valve replacement — performed through a ministernotomy or right anterior minithoracotomy with thoracoscopic or robotic assistance — has further narrowed the patient experience differential between surgical and transcatheter approaches, reducing incision length, postoperative pain, hospital length of stay, and recovery time compared to the full median sternotomy of earlier surgical eras.
The Heart Team discussion — the multidisciplinary evaluation by cardiac surgery, interventional cardiology, cardiac imaging, and anesthesia that is now recommended by guidelines as the standard decision-making process for aortic stenosis management — exists precisely to navigate the complexity of these trade-offs. For the fifty-five-year-old patient with severe symptomatic aortic stenosis and low surgical risk who presents to a Heart Team discussion, the conversation should not default to TAVR because it is technically available and procedurally appealing. It should engage honestly with the durability data, the reintervention landscape, the patient's individual anatomy, lifestyle, anticoagulation tolerance, and long-term expectations — and arrive at a recommendation that serves the patient's interests across their entire expected lifetime, not merely across the next two to three years of trial follow-up.
Surgical aortic valve replacement in low-risk patients, particularly at experienced centers with high surgical volumes, carries thirty-day mortality rates of well under one percent and stroke rates of one to two percent — outcomes that are equivalent to or approaching those of TAVR in the same risk category, and that are achieved with the implantation of a prosthesis whose durability profile is substantially better defined and longer-established than that of any currently available TAVR device. The modern era of minimally invasive surgical aortic valve replacement — performed through a ministernotomy or right anterior minithoracotomy with thoracoscopic or robotic assistance — has further narrowed the patient experience differential between surgical and transcatheter approaches, reducing incision length, postoperative pain, hospital length of stay, and recovery time compared to the full median sternotomy of earlier surgical eras.
The Heart Team discussion — the multidisciplinary evaluation by cardiac surgery, interventional cardiology, cardiac imaging, and anesthesia that is now recommended by guidelines as the standard decision-making process for aortic stenosis management — exists precisely to navigate the complexity of these trade-offs. For the fifty-five-year-old patient with severe symptomatic aortic stenosis and low surgical risk who presents to a Heart Team discussion, the conversation should not default to TAVR because it is technically available and procedurally appealing. It should engage honestly with the durability data, the reintervention landscape, the patient's individual anatomy, lifestyle, anticoagulation tolerance, and long-term expectations — and arrive at a recommendation that serves the patient's interests across their entire expected lifetime, not merely across the next two to three years of trial follow-up.
The Patient's Perspective: What Younger Patients Are Actually Deciding
The shift in younger patients' decision-making toward surgical aortic valve replacement is not a theoretical projection — it is a clinical reality that is beginning to be documented in registry data and shared decision-making literature. Younger patients who have been fully informed about the durability limitations of transcatheter bioprostheses and the complexity of valve-in-valve reintervention frequently and rationally choose the more invasive upfront option when the long-term arithmetic is laid out transparently: one more demanding recovery now, in exchange for the near-certainty of a valve that will not require replacement during their active working and family years.
This decision reflects a different temporal horizon than the one that governed early TAVR decision-making. The elderly, frail patient for whom TAVR was designed is optimizing for survival and quality of life in the near to medium term. The fifty-year-old professional who plays tennis, expects to work for another fifteen years, and has two children in school is optimizing across a fundamentally different time horizon — one in which the prospect of a complex, high-risk redo procedure at sixty-two, with uncertain coronary anatomy and a deteriorated valve body, is a materially worse outcome than an open surgical procedure performed electively today while surgical risk is at its nadir.
The communication challenge for clinicians is substantial. Patients presenting for aortic valve evaluation in their fifties are frequently in the grip of a powerful cognitive bias toward the less invasive option — the human tendency to weight immediate pain and risk more heavily than future risk of equal or greater magnitude. Helping patients overcome this bias requires not simply presenting the data but framing it in terms that are meaningful at their life stage: not "the valve has an eighty percent chance of surviving to ten years" but "there is a one in five chance that you will need another valve procedure before your younger child finishes university."
This decision reflects a different temporal horizon than the one that governed early TAVR decision-making. The elderly, frail patient for whom TAVR was designed is optimizing for survival and quality of life in the near to medium term. The fifty-year-old professional who plays tennis, expects to work for another fifteen years, and has two children in school is optimizing across a fundamentally different time horizon — one in which the prospect of a complex, high-risk redo procedure at sixty-two, with uncertain coronary anatomy and a deteriorated valve body, is a materially worse outcome than an open surgical procedure performed electively today while surgical risk is at its nadir.
The communication challenge for clinicians is substantial. Patients presenting for aortic valve evaluation in their fifties are frequently in the grip of a powerful cognitive bias toward the less invasive option — the human tendency to weight immediate pain and risk more heavily than future risk of equal or greater magnitude. Helping patients overcome this bias requires not simply presenting the data but framing it in terms that are meaningful at their life stage: not "the valve has an eighty percent chance of surviving to ten years" but "there is a one in five chance that you will need another valve procedure before your younger child finishes university."
Emerging Technologies: The Next Generation of Durable Transcatheter Valves
It would be incomplete to discuss the durability limitations of current TAVR prostheses without acknowledging the active and well-funded effort underway to address them. Several strategies are in preclinical or early clinical development to improve transcatheter valve longevity: anti-calcification treatments applied to bioprosthetic tissue during manufacture, polymer leaflet valves that eliminate the biological tissue degradation pathway entirely, tissue engineering approaches using decellularized scaffolds that may allow recellularization and biological integration after implantation, and mechanical transcatheter valves that avoid bioprosthetic failure modes altogether.
Each of these approaches holds genuine promise, and the pace of innovation in the transcatheter valve space has been extraordinary. However, the clinical validation of durability claims for novel valve technologies requires decades of follow-up in human implantation studies — a timeline that cannot be compressed by engineering innovation alone. A younger patient deciding between TAVR and surgical aortic valve replacement today cannot make that decision on the basis of durability data that will not be available for fifteen years. They must decide on the basis of what is known now, using the best available evidence about currently implantable devices.
The practical clinical implication is that the emergence of a more durable transcatheter option, when it arrives with adequate long-term evidence, will likely shift the decision calculus for younger patients substantially. Until that evidence exists, the surgical option — whether mechanical or biological through open surgery, or the Ross procedure in selected centers — retains a durability advantage that is supported by decades of follow-up data and that the transcatheter alternatives cannot yet match.
Each of these approaches holds genuine promise, and the pace of innovation in the transcatheter valve space has been extraordinary. However, the clinical validation of durability claims for novel valve technologies requires decades of follow-up in human implantation studies — a timeline that cannot be compressed by engineering innovation alone. A younger patient deciding between TAVR and surgical aortic valve replacement today cannot make that decision on the basis of durability data that will not be available for fifteen years. They must decide on the basis of what is known now, using the best available evidence about currently implantable devices.
The practical clinical implication is that the emergence of a more durable transcatheter option, when it arrives with adequate long-term evidence, will likely shift the decision calculus for younger patients substantially. Until that evidence exists, the surgical option — whether mechanical or biological through open surgery, or the Ross procedure in selected centers — retains a durability advantage that is supported by decades of follow-up data and that the transcatheter alternatives cannot yet match.
Special Considerations: Women, Athletes, and Patients Planning Pregnancy
Several patient subgroups within the younger aortic stenosis population warrant specific consideration in the valve selection discussion. Women of childbearing age who wish to conceive following valve replacement face a particular dilemma: warfarin anticoagulation — required for mechanical valve prostheses — is teratogenic in the first trimester, necessitating either conversion to low-molecular-weight heparin during the period of organogenesis (carrying an increased maternal thromboembolic risk) or avoidance of mechanical valve selection in women who intend future pregnancy. Biological bioprosthetic valves allow safer pregnancy management with anticoagulation limited to low-molecular-weight heparin throughout gestation, but the hemodynamic demands of pregnancy — increased cardiac output, heart rate, and blood volume — accelerate bioprosthetic structural deterioration, meaning that the durability of a biological valve implanted in a woman of reproductive age is further reduced by the hemodynamic stress of pregnancy.
Young athletes and physically active patients present the valve selection question from a different angle: the concern that warfarin anticoagulation in a patient who participates in contact sports or high-risk recreational activities carries an unacceptable bleeding risk from traumatic injuries. For these patients, the anticoagulation burden of mechanical valve selection must be weighed against their specific activity profile, and the biological option — despite its durability limitations — may represent the better individualized choice, particularly if complemented by a clear plan for surveillance and timely reintervention.
These nuances reinforce the central argument for individualized, multidisciplinary decision-making in younger patients with aortic stenosis. There is no single correct answer to the valve selection question for patients under sixty — there is only a process of careful, transparent, patient-centered discussion that weighs the full spectrum of considerations relevant to that individual's life circumstances, expectations, and preferences.
Young athletes and physically active patients present the valve selection question from a different angle: the concern that warfarin anticoagulation in a patient who participates in contact sports or high-risk recreational activities carries an unacceptable bleeding risk from traumatic injuries. For these patients, the anticoagulation burden of mechanical valve selection must be weighed against their specific activity profile, and the biological option — despite its durability limitations — may represent the better individualized choice, particularly if complemented by a clear plan for surveillance and timely reintervention.
These nuances reinforce the central argument for individualized, multidisciplinary decision-making in younger patients with aortic stenosis. There is no single correct answer to the valve selection question for patients under sixty — there is only a process of careful, transparent, patient-centered discussion that weighs the full spectrum of considerations relevant to that individual's life circumstances, expectations, and preferences.
Conclusion: Ten Years Is Not a Lifetime
The surgical hands in Figure 1 — steady, precise, operating within the opened field of a living heart — represent a choice. Not a failure of technology, not a retreat from innovation, but a deliberate and considered clinical decision that the open surgical approach, with all its immediate demands on patient tolerance and recovery, provides something that the catheter cannot yet offer to a younger patient: a valve whose durability horizon is measured in decades rather than years.
The narrative of TAVR has been, appropriately, a narrative of expansion — from the inoperable patient for whom it was a last resort, to the high-risk patient for whom it offered an alternative to prohibitive surgical risk, to the intermediate and low-risk patient in whom it demonstrated equivalent short-term outcomes. Each step of that expansion has been evidence-based, and each step has benefited patients who were well matched to the procedure's profile. The current frontier — the young, low-risk patient with aortic stenosis and a forty-year life expectancy — tests the procedure against a different standard, and it is a standard that the current generation of transcatheter prostheses does not yet meet.
The clinicians who are having the more difficult, more honest, more temporally complete conversation with their younger patients — who are presenting the ten-year durability data without minimizing it, who are explaining valve-in-valve reintervention and its coronary obstruction risk without eliding its complexity, and who are offering surgical aortic valve replacement as the considered first choice rather than the fallback option — are not being reactionary. They are being precise. Ten years, as they and their patients are increasingly understanding, is not a lifetime. And for a fifty-year-old with a new valve, a lifetime is exactly what the decision demands to serve.
The narrative of TAVR has been, appropriately, a narrative of expansion — from the inoperable patient for whom it was a last resort, to the high-risk patient for whom it offered an alternative to prohibitive surgical risk, to the intermediate and low-risk patient in whom it demonstrated equivalent short-term outcomes. Each step of that expansion has been evidence-based, and each step has benefited patients who were well matched to the procedure's profile. The current frontier — the young, low-risk patient with aortic stenosis and a forty-year life expectancy — tests the procedure against a different standard, and it is a standard that the current generation of transcatheter prostheses does not yet meet.
The clinicians who are having the more difficult, more honest, more temporally complete conversation with their younger patients — who are presenting the ten-year durability data without minimizing it, who are explaining valve-in-valve reintervention and its coronary obstruction risk without eliding its complexity, and who are offering surgical aortic valve replacement as the considered first choice rather than the fallback option — are not being reactionary. They are being precise. Ten years, as they and their patients are increasingly understanding, is not a lifetime. And for a fifty-year-old with a new valve, a lifetime is exactly what the decision demands to serve.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis