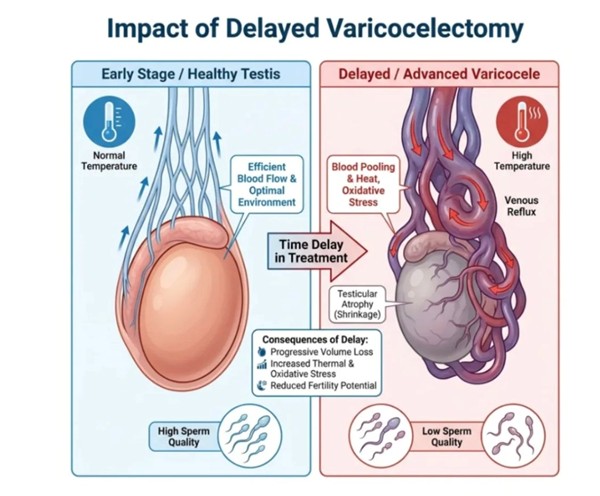
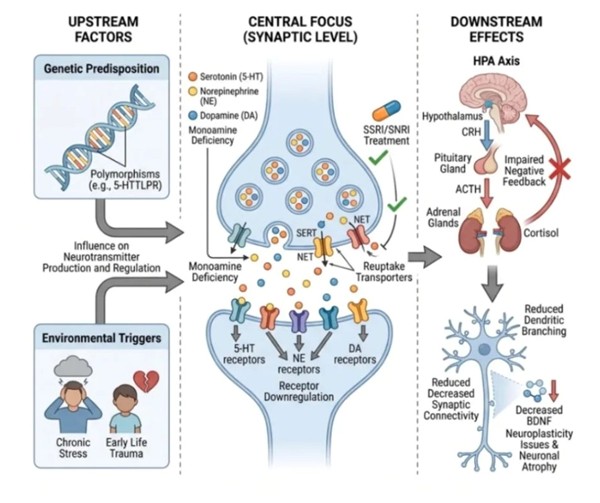
-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
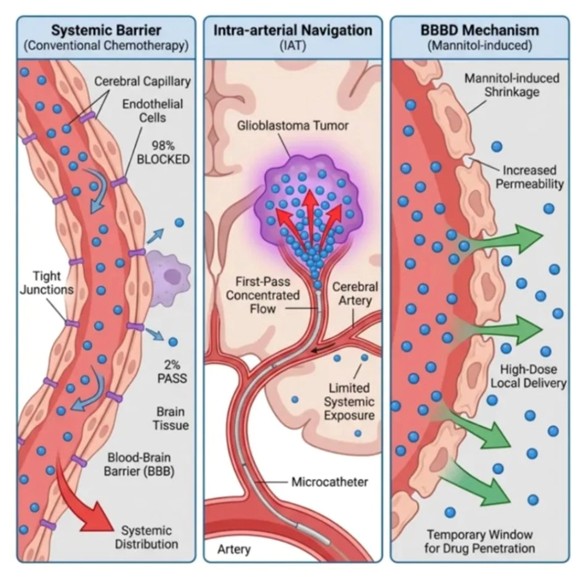
Rewiring the Route: How Intra-arterial Therapy Outsmarts the Blood-Brain Barrier in Brain Tumor Treatment
The Fortress That Protects — and Obstructs: Understanding the Blood-Brain Barrier
The human brain is one of the most privileged organs in the body, shielded from the outside world by a remarkable biological structure called the Blood-Brain Barrier. This barrier is formed by the highly specialized endothelial cells that line the brain's cerebral capillaries, reinforced by protein complexes known as tight junctions that seal the microscopic gaps between adjacent cells. The result is a near-impenetrable wall designed to selectively allow only essential nutrients, gases, and small lipid-soluble molecules into brain tissue while actively excluding pathogens, toxins, and — critically for oncology — most pharmaceutical agents. When conventional chemotherapy is administered intravenously, the drug becomes diluted throughout the body's entire circulatory volume, and upon reaching the cerebral vasculature, approximately 98% of the drug is blocked by these tight junctions and swept away into systemic distribution. Only a residual 2% manages to cross into the brain parenchyma — a fraction so small it is often therapeutically inadequate against an aggressive tumor like glioblastoma multiforme, while the majority of the drug continues to circulate systemically, contributing to the well-documented side effects of conventional chemotherapy without proportionate benefit to the tumor site.
The Direct Route: Intra-arterial Therapy and the First-Pass Advantage
Intra-arterial Therapy addresses the fundamental inefficiency of systemic delivery not by attempting to improve drug penetration across the BBB globally, but by eliminating the need for widespread distribution altogether. Using real-time fluoroscopic guidance, an interventional neuroradiologist threads a flexible microcatheter through the femoral artery, advancing it with precision through the vascular tree until its tip is positioned directly within the specific cerebral artery that supplies blood to the tumor-bearing region of the brain. The chemotherapeutic agent is then infused at this precise anatomical location. This technique exploits what is known as the first-pass effect — before the drug has any opportunity to be diluted by systemic recirculation, it arrives at the tumor in a highly concentrated bolus. The clinical implications are profound: drug concentrations at the tumor site can be an order of magnitude higher than those achievable through intravenous administration, while systemic exposure — and therefore systemic toxicity — is substantially reduced. For patients with glioblastoma, whose tumors are supplied by identifiable cerebral arteries, IAT offers a targeted pharmacological intervention that aligns the delivery mechanism with the biology of the disease.
Hacking the Barrier: Blood-Brain Barrier Disruption with Mannitol
Even with the first-pass advantage of IAT, the residual integrity of the Blood-Brain Barrier at the tumor's margin can still restrict the volume of drug that ultimately reaches infiltrating tumor cells. To overcome this final obstacle, clinicians can deploy an additional technique: Blood-Brain Barrier Disruption, most commonly achieved using a hyperosmolar solution of mannitol infused intra-arterially before the chemotherapeutic agent. When mannitol enters the cerebral vasculature, it creates a sharp osmotic gradient between the blood and the endothelial cells lining the capillaries. In response, water is rapidly drawn out of the endothelial cells into the bloodstream, causing them to shrink. This cellular shrinkage physically widens the tight junctions, dramatically increasing the permeability of the barrier and creating a temporary window — typically lasting between 15 and 30 minutes — through which large-molecule drugs that would ordinarily be excluded can now penetrate the brain tissue in high-dose concentrations. The therapeutic implications of combining IAT with BBBD are significant: the technique allows not only for concentrated local delivery via the intra-arterial route, but also for maximal tissue penetration at and beyond the tumor margin, targeting the invasive leading edge of glioblastoma that conventional surgery and systemic therapy routinely fail to reach.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis