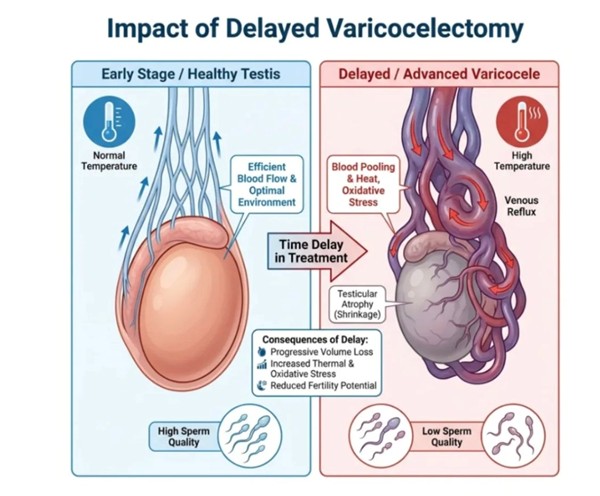
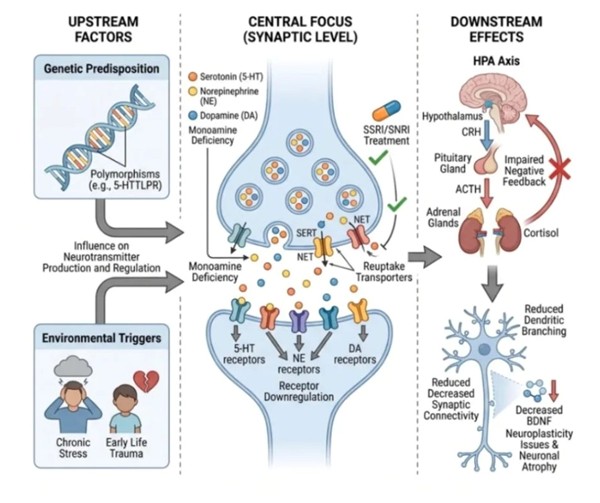
-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
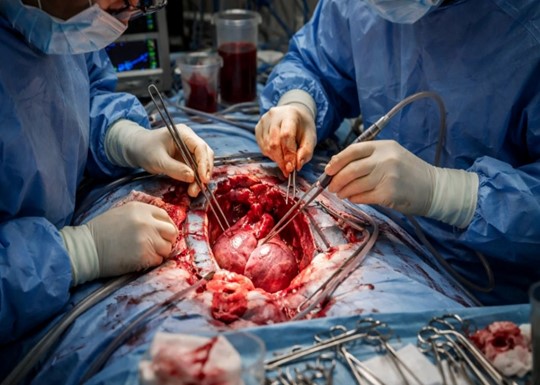
Figure 1. An open cardiothoracic surgical field, where multiple surgeons navigate complex exposed tissue with precision instruments under high-stakes conditions. This image captures the irreducible reality of operative surgery — the environment of zero margin for error that makes pre-operative rehearsal through AI-generated digital twins not merely a technological convenience but a patient safety imperative.
Rehearse Before You Cut: AI-Generated Digital Twins and the Future of Pre-Operative Surgical Planning
Before the First Incision
Every surgeon who has stood at an operating table knows the particular quality of the moment before the first incision. The preoperative imaging has been reviewed, the team briefed, the instruments counted and arranged. The surgical plan exists as a mental model — a three-dimensional cognitive map assembled from two-dimensional scans, from pathology reports, from the pattern recognition of prior cases, and from the surgeon's accumulated experience with the anatomy of the human body in general. What it does not contain, in most operating rooms today, is a precise simulation of this patient's specific anatomy, the specific challenges their physiology will present, or the specific decisions that will need to be made when the tissue planes diverge from expectation.
That gap between the generic anatomical model and the specific patient in front of the surgeon is where errors live. It is where the unexpected vessel variant produces unexpected hemorrhage, where the tumor adhesion to a structure that imaging suggested was free-standing requires a decision that was not rehearsed, where the anatomical landmark that orients the surgeon in textbook cases is absent or distorted by prior surgery, disease, or individual variation. Intraoperative surprises, in surgery as in most high-stakes domains, are not merely inconvenient. They are dangerous.
The promise of AI-generated digital twins in surgical planning is the systematic reduction of that gap. By constructing a computationally accurate, patient-specific virtual replica of the relevant anatomy from preoperative imaging data, and by using artificial intelligence to simulate the tissue mechanics, vascular behavior, and physiological responses that the surgical intervention will provoke, the digital twin enables the surgical team to rehearse the specific procedure on the specific patient before making a single real incision. The operating room depicted in Figure 1 — its intensity, its complexity, its absolute requirement for precision under pressure — is why that rehearsal matters.
That gap between the generic anatomical model and the specific patient in front of the surgeon is where errors live. It is where the unexpected vessel variant produces unexpected hemorrhage, where the tumor adhesion to a structure that imaging suggested was free-standing requires a decision that was not rehearsed, where the anatomical landmark that orients the surgeon in textbook cases is absent or distorted by prior surgery, disease, or individual variation. Intraoperative surprises, in surgery as in most high-stakes domains, are not merely inconvenient. They are dangerous.
The promise of AI-generated digital twins in surgical planning is the systematic reduction of that gap. By constructing a computationally accurate, patient-specific virtual replica of the relevant anatomy from preoperative imaging data, and by using artificial intelligence to simulate the tissue mechanics, vascular behavior, and physiological responses that the surgical intervention will provoke, the digital twin enables the surgical team to rehearse the specific procedure on the specific patient before making a single real incision. The operating room depicted in Figure 1 — its intensity, its complexity, its absolute requirement for precision under pressure — is why that rehearsal matters.
The Digital Twin: Concept, Construction, and Computational Foundation From Engineering to Medicine: The Origin of the Digital Twin Concept
The concept of the digital twin originated not in medicine but in aerospace engineering, where NASA and defense contractors developed the practice of creating virtual replicas of physical systems — aircraft, spacecraft, propulsion systems — that could be continuously updated with real-world sensor data and used to simulate performance under conditions that were too dangerous, too expensive, or too time-consuming to test physically. A digital twin of a jet engine, in this context, is not a static three-dimensional model but a dynamically updated computational representation that captures the engine's current state, predicts its behavior under specified operational conditions, and identifies failure modes before they manifest in the physical system.
The translation of this concept into surgical planning follows the same underlying logic. A patient's anatomy is a physical system — geometrically complex, mechanically active, physiologically dynamic — that can, in principle, be replicated in silico with sufficient fidelity to predict its behavior under the mechanical perturbations of surgical intervention. The obstacles to this translation are formidable: biological tissue is far more complex, heterogeneous, and variable than engineered materials; the imaging modalities available for capturing anatomical geometry introduce resolution limitations and artifacts; and the computational models required to simulate tissue mechanics, fluid dynamics, and physiological response are orders of magnitude more complex than those used for engineered systems.
Artificial intelligence, and specifically the deep learning architectures that have transformed medical image analysis over the past decade, has provided the tools to overcome these obstacles at a level of fidelity that was not achievable with earlier computational approaches. The digital twin in its surgical planning incarnation is therefore better understood as an AI-augmented computational model than as a simple three-dimensional reconstruction — a model that uses machine learning to estimate tissue properties, predict deformation under load, simulate fluid dynamics within vascular structures, and generate probabilistic maps of anatomical risk that go far beyond the static visualization of conventional preoperative imaging.
The translation of this concept into surgical planning follows the same underlying logic. A patient's anatomy is a physical system — geometrically complex, mechanically active, physiologically dynamic — that can, in principle, be replicated in silico with sufficient fidelity to predict its behavior under the mechanical perturbations of surgical intervention. The obstacles to this translation are formidable: biological tissue is far more complex, heterogeneous, and variable than engineered materials; the imaging modalities available for capturing anatomical geometry introduce resolution limitations and artifacts; and the computational models required to simulate tissue mechanics, fluid dynamics, and physiological response are orders of magnitude more complex than those used for engineered systems.
Artificial intelligence, and specifically the deep learning architectures that have transformed medical image analysis over the past decade, has provided the tools to overcome these obstacles at a level of fidelity that was not achievable with earlier computational approaches. The digital twin in its surgical planning incarnation is therefore better understood as an AI-augmented computational model than as a simple three-dimensional reconstruction — a model that uses machine learning to estimate tissue properties, predict deformation under load, simulate fluid dynamics within vascular structures, and generate probabilistic maps of anatomical risk that go far beyond the static visualization of conventional preoperative imaging.
Construction Pipeline: From Imaging Data to Virtual Patient
The construction of a patient-specific surgical digital twin begins with the acquisition of high-resolution cross-sectional imaging — typically computed tomography angiography for vascular and cardiac structures, magnetic resonance imaging for soft tissue characterization, or a multimodal combination that captures both the geometric and the compositional properties of the relevant anatomy. The raw imaging data is then processed through an AI-driven segmentation pipeline that assigns tissue labels to each voxel of the imaging volume, distinguishing arterial wall from lumen, tumor from parenchyma, nerve from connective tissue, and bone from cartilage with a precision and consistency that human manual segmentation cannot match at clinical scale.
The segmented data is then used to construct a three-dimensional geometric mesh — a mathematical representation of the anatomical surfaces that can be rendered visually, manipulated spatially, and used as the geometric substrate for biomechanical simulation. This mesh is not a passive visualization tool; it is the foundation upon which computational models of tissue mechanics are applied. Finite element analysis methods, long established in engineering for simulating stress distribution in complex structures, are applied to the anatomical mesh to predict how tissue will deform under the forces of surgical retraction, how vessel walls will respond to clamping or suturing, and how organ parenchyma will behave when manipulated or incised.
Complementing the structural mechanical models are computational fluid dynamics simulations that characterize blood flow patterns, pressure gradients, and shear stress distributions within the vascular anatomy of interest. For cardiac and vascular surgical planning, these hemodynamic simulations are particularly critical: they reveal where flow turbulence will develop after a proposed anastomosis, what pressure gradients will exist across a reconstruction, and how the vascular network will redistribute flow following ligation or bypass of specific vessels. These are precisely the predictions that cannot be made from static imaging alone and that have historically been accessible only through the surgeon's pattern-matched intuition.
The segmented data is then used to construct a three-dimensional geometric mesh — a mathematical representation of the anatomical surfaces that can be rendered visually, manipulated spatially, and used as the geometric substrate for biomechanical simulation. This mesh is not a passive visualization tool; it is the foundation upon which computational models of tissue mechanics are applied. Finite element analysis methods, long established in engineering for simulating stress distribution in complex structures, are applied to the anatomical mesh to predict how tissue will deform under the forces of surgical retraction, how vessel walls will respond to clamping or suturing, and how organ parenchyma will behave when manipulated or incised.
Complementing the structural mechanical models are computational fluid dynamics simulations that characterize blood flow patterns, pressure gradients, and shear stress distributions within the vascular anatomy of interest. For cardiac and vascular surgical planning, these hemodynamic simulations are particularly critical: they reveal where flow turbulence will develop after a proposed anastomosis, what pressure gradients will exist across a reconstruction, and how the vascular network will redistribute flow following ligation or bypass of specific vessels. These are precisely the predictions that cannot be made from static imaging alone and that have historically been accessible only through the surgeon's pattern-matched intuition.
Rehearsal in the Virtual Operating Room: What Simulation Offers That Planning Meetings Cannot
The distinction between traditional preoperative planning and digital twin-enabled surgical rehearsal is not merely technical — it is epistemological. Traditional planning is a cognitive exercise: the surgeon reviews imaging, constructs a mental model, and develops a plan that exists as a sequence of intended actions. The plan is evaluated against the surgeon's prior experience, their knowledge of anatomical variation, and their judgment about which contingencies are most likely. It is, in a fundamental sense, a map drawn from memory overlaid on a photograph of unfamiliar terrain.
Digital twin-based surgical rehearsal is an empirical exercise: the surgeon performs the procedure — virtually, on the specific patient's anatomy — before performing it physically. The virtual execution exposes the plan to the specific anatomical realities of the individual case in a way that cognitive review cannot. The retractor placed at a specific angle in the virtual model either provides adequate exposure or it does not — and the simulation tells the surgeon which, before the patient is positioned on the operating table. The proposed incision line for a liver resection either achieves the planned oncological margin while preserving adequate functional parenchyma, or it does not — and the digital twin reveals the discrepancy before any tissue is divided.
Digital twin-based surgical rehearsal is an empirical exercise: the surgeon performs the procedure — virtually, on the specific patient's anatomy — before performing it physically. The virtual execution exposes the plan to the specific anatomical realities of the individual case in a way that cognitive review cannot. The retractor placed at a specific angle in the virtual model either provides adequate exposure or it does not — and the simulation tells the surgeon which, before the patient is positioned on the operating table. The proposed incision line for a liver resection either achieves the planned oncological margin while preserving adequate functional parenchyma, or it does not — and the digital twin reveals the discrepancy before any tissue is divided.
Trajectory Planning and Exposure Optimization
One of the most practically consequential applications of surgical digital twins is the optimization of operative trajectories — the paths through tissue that instruments must travel to reach the surgical target, and the spatial configuration of retractors, suction devices, and surgical hands that are required to maintain adequate visualization throughout the procedure. In deep or anatomically complex surgical fields — the posterior mediastinum, the retroperitoneum, the base of skull, the deep pelvis — trajectory planning is among the most technically demanding and consequential aspects of operative decision-making, and errors in trajectory selection produce complications that include injury to adjacent neurovascular structures, inadequate tumor exposure requiring conversion to a more morbid approach, and fatigue-inducing ergonomic positions that increase the risk of fine motor error as the case progresses.
Digital twin simulation allows the surgeon to model multiple trajectory options in the virtual environment, evaluating each for the quality of surgical field exposure it provides, the structures it places at risk along its path, and the instrument configurations it requires. This systematic evaluation — performed at leisure in the simulation environment rather than under time pressure in the operating room — produces a trajectory selection that is grounded in the specific geometry of the individual case rather than the generic guidance of anatomical atlases. For minimally invasive approaches, where port placement determines the mechanical workspace available for the entire procedure, this preoperative trajectory optimization is particularly valuable, as errors in port placement cannot be corrected without repositioning that adds operative time and anesthetic exposure.
Digital twin simulation allows the surgeon to model multiple trajectory options in the virtual environment, evaluating each for the quality of surgical field exposure it provides, the structures it places at risk along its path, and the instrument configurations it requires. This systematic evaluation — performed at leisure in the simulation environment rather than under time pressure in the operating room — produces a trajectory selection that is grounded in the specific geometry of the individual case rather than the generic guidance of anatomical atlases. For minimally invasive approaches, where port placement determines the mechanical workspace available for the entire procedure, this preoperative trajectory optimization is particularly valuable, as errors in port placement cannot be corrected without repositioning that adds operative time and anesthetic exposure.
Anomaly Detection and Contingency Planning
Among the highest-value applications of AI-powered surgical digital twins is the systematic identification of anatomical anomalies and the development of contingency plans for the scenarios they create. Anatomical variation — aberrant arterial origins, anomalous venous drainage patterns, unusual nerve trajectories, unexpected adhesions from prior disease or surgery — is far more common than the surgical trainee's education on standard anatomy suggests. Published studies of operative anatomy consistently document significant variation from textbook descriptions in twenty to forty percent of cases across multiple organ systems, and it is precisely these variant cases in which surgical complications are most likely to occur.
AI-driven analysis of the digital twin can flag anatomical features that deviate from population norms and quantify the clinical significance of those deviations for the planned procedure. A hepatic arterial variant that would not be visible on a standard preoperative CT review — but that would be encountered as a life-threatening hemorrhage if divided during a laparoscopic cholecystectomy — is identifiable in the digital twin analysis and can be incorporated into the operative plan as a specific anatomical landmark to be protected. The surgical team can rehearse the dissection around this variant vessel in the simulation environment, developing the muscle memory and decision protocols for its management before they encounter it in tissue.
Contingency planning — the preparation of decision trees for the scenarios that are possible but not certain — is enhanced by the digital twin's ability to simulate alternative anatomical states and operative scenarios in parallel. The surgeon can virtually execute both the planned approach and the contingency approach, identifying the decision point at which one would be abandoned for the other, and establishing the visual or tactile cues that will signal when that decision point has been reached in the real case. This systematic contingency rehearsal converts the intraoperative “surprise” from a crisis requiring improvised response into a recognized scenario with a pre-rehearsed management protocol.
AI-driven analysis of the digital twin can flag anatomical features that deviate from population norms and quantify the clinical significance of those deviations for the planned procedure. A hepatic arterial variant that would not be visible on a standard preoperative CT review — but that would be encountered as a life-threatening hemorrhage if divided during a laparoscopic cholecystectomy — is identifiable in the digital twin analysis and can be incorporated into the operative plan as a specific anatomical landmark to be protected. The surgical team can rehearse the dissection around this variant vessel in the simulation environment, developing the muscle memory and decision protocols for its management before they encounter it in tissue.
Contingency planning — the preparation of decision trees for the scenarios that are possible but not certain — is enhanced by the digital twin's ability to simulate alternative anatomical states and operative scenarios in parallel. The surgeon can virtually execute both the planned approach and the contingency approach, identifying the decision point at which one would be abandoned for the other, and establishing the visual or tactile cues that will signal when that decision point has been reached in the real case. This systematic contingency rehearsal converts the intraoperative “surprise” from a crisis requiring improvised response into a recognized scenario with a pre-rehearsed management protocol.
Clinical Applications: Where Digital Twins Are Demonstrating Real-World Impact Cardiac and Vascular Surgery: The Hemodynamic Simulation Frontier
Cardiac and vascular surgery represent the domains where computational digital twins have the most mature evidence base and the most directly demonstrable clinical impact. The geometry of the heart and great vessels is captured with high fidelity by modern CT angiography, and the physics of cardiac mechanics and vascular hemodynamics are sufficiently well characterized that finite element and computational fluid dynamics models can generate predictions with clinical relevance. For aortic root surgery, digital twin simulation of the planned reconstruction allows the surgeon to evaluate the hemodynamic consequences of different annuloplasty ring sizes, different valve reimplantation geometries, and different ascending aorta replacement lengths — decisions that are conventionally made intraoperatively on the basis of direct visual inspection and intraoperative echocardiography, but that can now be informed by preoperative simulation of their hemodynamic consequences.
For complex congenital heart disease, where the anatomical variability between patients can be extreme and standard operative templates are often inapplicable, digital twin-based planning has demonstrated particular clinical value. The heterotaxy syndromes, the complex single-ventricle anatomies, the rare conotruncal malformations that are encountered only occasionally even at the most experienced pediatric cardiac centers — these are precisely the cases where a patient-specific virtual model, rehearsed before the chest is opened, can provide the operating team with a level of anatomical familiarity that cannot be acquired from prior case experience alone.
For complex congenital heart disease, where the anatomical variability between patients can be extreme and standard operative templates are often inapplicable, digital twin-based planning has demonstrated particular clinical value. The heterotaxy syndromes, the complex single-ventricle anatomies, the rare conotruncal malformations that are encountered only occasionally even at the most experienced pediatric cardiac centers — these are precisely the cases where a patient-specific virtual model, rehearsed before the chest is opened, can provide the operating team with a level of anatomical familiarity that cannot be acquired from prior case experience alone.
Oncological Surgery: Margin Planning and Parenchymal Preservation
In oncological surgery, digital twin simulation addresses the fundamental tension between oncological radicality and functional preservation — the competing imperatives of removing all malignant tissue with adequate margins and preserving sufficient functional organ parenchyma to maintain post-resection quality of life. For hepatic surgery, where the relationship between the tumor, the hepatic vascular anatomy, and the portal and biliary segmental anatomy determines both the technical feasibility of resection and the functional adequacy of the remnant liver, virtual hepatectomy planning using patient-specific models has demonstrated reductions in post-resection liver failure, better margin achievement, and more consistent oncological outcomes compared to conventional anatomical planning.
For pulmonary surgery, where the identification of intersegmental planes for anatomical segmentectomy — a parenchyma-sparing alternative to lobectomy for early-stage lung cancer — requires precise understanding of the individual patient’s segmental bronchial and vascular branching patterns, digital twin models derived from chest CT allow the surgeon to plan the resection with full knowledge of the specific anatomy they will encounter at thoracoscopy, substantially reducing the risk of incomplete intersegmental plane identification that leads to positive margins or unintended parenchymal loss.
For pulmonary surgery, where the identification of intersegmental planes for anatomical segmentectomy — a parenchyma-sparing alternative to lobectomy for early-stage lung cancer — requires precise understanding of the individual patient’s segmental bronchial and vascular branching patterns, digital twin models derived from chest CT allow the surgeon to plan the resection with full knowledge of the specific anatomy they will encounter at thoracoscopy, substantially reducing the risk of incomplete intersegmental plane identification that leads to positive margins or unintended parenchymal loss.
Neurosurgery and Skull Base Surgery: Millimeter-Level Planning in Critical Anatomy
Nowhere in surgery is the margin for anatomical error smaller than in neurosurgery and skull base surgery, where the relevant structures — the cranial nerves, the cerebral vasculature, the eloquent cortical areas — are measured in millimeters and where a deviation of two or three millimeters from the planned trajectory can mean the difference between a radical tumor resection and a life-altering neurological deficit. Digital twin simulation in this context does not merely improve surgical efficiency; it defines the operational boundary between what is oncologically achievable and what is neurologically safe for the individual patient.
AI-driven tractography — the computational mapping of white matter fiber tracts from diffusion tensor imaging data — integrated into the digital twin allows the neurosurgeon to visualize the relationship between the planned surgical corridor and the eloquent white matter pathways that run adjacent to it, identifying the approach that achieves maximum tumor access while minimizing eloquent white matter disruption. Combined with functional MRI mapping of cortical eloquent areas and intraoperative neuronavigation registration, the preoperative digital twin becomes the navigational substrate for the entire procedure, translating the rehearsed virtual approach into real-time guidance within the patient-specific anatomy.
AI-driven tractography — the computational mapping of white matter fiber tracts from diffusion tensor imaging data — integrated into the digital twin allows the neurosurgeon to visualize the relationship between the planned surgical corridor and the eloquent white matter pathways that run adjacent to it, identifying the approach that achieves maximum tumor access while minimizing eloquent white matter disruption. Combined with functional MRI mapping of cortical eloquent areas and intraoperative neuronavigation registration, the preoperative digital twin becomes the navigational substrate for the entire procedure, translating the rehearsed virtual approach into real-time guidance within the patient-specific anatomy.
The Role of AI: Beyond Visualization to Prediction
The distinction between a digital twin and a conventional three-dimensional reconstruction lies in the AI layer that animates the model. A three-dimensional reconstruction renders anatomical geometry in a visually intuitive form — valuable, but ultimately a passive display of information already present in the imaging data. A digital twin, as AI-augmented, is a predictive instrument: it generates information that is not directly present in the imaging data but is inferred from it through computational models of tissue behavior, fluid dynamics, and physiological response.
The predictive capabilities of modern surgical digital twins include tissue deformation prediction — how the brain will shift after craniotomy and dural opening, altering the correspondence between preoperative imaging and intraoperative anatomy; vascular response modeling — how a vessel will behave when clamped, mobilized, or subjected to the tension of an anastomosis under physiological pressure; and tumor vascularity assessment — mapping the feeding vessels and internal vascular architecture of a tumor from contrast enhancement patterns in a way that predicts intraoperative bleeding risk and guides dissection strategy.
The generation of risk maps — probabilistic visual overlays on the anatomical model that assign quantified risk scores to different regions of the surgical field based on proximity to critical structures, tissue vulnerability, and the mechanical demands of planned maneuvers — represents one of the most clinically actionable outputs of AI-driven digital twin analysis. A risk map that highlights the region around an aberrant hepatic artery in a cholecystectomy, or the segment of aorta adjacent to a tumor in a retroperitoneal dissection, provides the surgeon with a spatially explicit, patient-specific caution that conventional anatomical knowledge cannot supply.
The predictive capabilities of modern surgical digital twins include tissue deformation prediction — how the brain will shift after craniotomy and dural opening, altering the correspondence between preoperative imaging and intraoperative anatomy; vascular response modeling — how a vessel will behave when clamped, mobilized, or subjected to the tension of an anastomosis under physiological pressure; and tumor vascularity assessment — mapping the feeding vessels and internal vascular architecture of a tumor from contrast enhancement patterns in a way that predicts intraoperative bleeding risk and guides dissection strategy.
The generation of risk maps — probabilistic visual overlays on the anatomical model that assign quantified risk scores to different regions of the surgical field based on proximity to critical structures, tissue vulnerability, and the mechanical demands of planned maneuvers — represents one of the most clinically actionable outputs of AI-driven digital twin analysis. A risk map that highlights the region around an aberrant hepatic artery in a cholecystectomy, or the segment of aorta adjacent to a tumor in a retroperitoneal dissection, provides the surgeon with a spatially explicit, patient-specific caution that conventional anatomical knowledge cannot supply.
Team Rehearsal and Communication: The Collective Benefit of a Shared Virtual Model
The benefits of surgical digital twins extend beyond the individual surgeon to the entire operating team. Complex surgical procedures — the multi-surgeon cardiac operations, the multi-specialty cancer resections, the vascular reconstructions requiring simultaneous cardiac and vascular operative strategies — demand a shared mental model among all team members: the operating surgeons, the anesthetist, the perfusionist, the scrub nurses, the surgical assistants. Divergences in team members’ understanding of the operative plan are a significant source of communication failures, instrument requests that cannot be fulfilled because the equipment was not anticipated, and handover errors at team transition points during long procedures.
The digital twin provides a shared visual object around which preoperative team rehearsal can be organized. Rather than a verbal description of the surgical plan — inherently limited by the difficulty of communicating three-dimensional spatial relationships through language alone — the team can review the virtual model together, walk through the planned procedure step by step in the simulation environment, and establish shared situational awareness of the anatomical challenges, the critical decision points, and the contingency protocols. This team-based rehearsal mirrors the pre-mission briefings of aviation and military special operations, domains in which the correlation between shared situational awareness and team performance under pressure is well established.
The anesthetic team benefits specifically from knowledge of the expected operative duration, the anticipated blood loss and fluid requirements, the likelihood of specific intraoperative events — aortic cross-clamping, deep hypothermic circulatory arrest, portal vein clamping — and their anticipated timing, allowing preparation of pharmacological and monitoring strategies that are calibrated to the specific physiological demands of the planned procedure rather than to generic protocols for the operative category.
The digital twin provides a shared visual object around which preoperative team rehearsal can be organized. Rather than a verbal description of the surgical plan — inherently limited by the difficulty of communicating three-dimensional spatial relationships through language alone — the team can review the virtual model together, walk through the planned procedure step by step in the simulation environment, and establish shared situational awareness of the anatomical challenges, the critical decision points, and the contingency protocols. This team-based rehearsal mirrors the pre-mission briefings of aviation and military special operations, domains in which the correlation between shared situational awareness and team performance under pressure is well established.
The anesthetic team benefits specifically from knowledge of the expected operative duration, the anticipated blood loss and fluid requirements, the likelihood of specific intraoperative events — aortic cross-clamping, deep hypothermic circulatory arrest, portal vein clamping — and their anticipated timing, allowing preparation of pharmacological and monitoring strategies that are calibrated to the specific physiological demands of the planned procedure rather than to generic protocols for the operative category.
Surgical Training and Education: The Simulation Layer
The educational implications of AI-generated surgical digital twins are, if anything, more consequential over the long term than their immediate clinical applications. Surgical training has historically depended on progressive exposure to operative cases under supervision — a model that is biologically sound in its principle of deliberate practice but that has always been limited by the ethical tension between the trainee’s learning needs and the patient’s right to receive care from the most skilled available operator. Work-hour restrictions, reduced surgical volumes in training programs, and the increasing complexity of index cases have intensified this tension in the contemporary training environment.
Digital twin simulation offers a path toward resolving this tension by allowing trainees to accumulate operative experience in a zero-risk virtual environment before applying their skills to real patients. Unlike the generic phantom models and cadaveric specimens that currently populate surgical simulation curricula — which provide valuable procedural exposure but cannot replicate the anatomical specificity of individual patient cases — patient-derived digital twins allow the trainee to practice the specific procedure on the specific anatomy they will encounter in the real case, with the specific challenges that the patient’s disease and anatomy present.
The integration of haptic feedback systems — devices that replicate the tactile resistance of biological tissue during virtual instrument interaction — with high-fidelity digital twin models is advancing rapidly, and the near-term prospect is of a training environment in which the trainee can feel the resistance of the virtual aortic wall under suture tension, or the compliance differential between tumor and normal parenchyma during dissection, in a way that approaches the proprioceptive richness of real operative experience. This haptic-augmented digital twin simulation is not a replacement for supervised operative training but a complement to it that allows trainees to enter supervised operative cases with a level of case-specific preparedness that has not previously been achievable.
Digital twin simulation offers a path toward resolving this tension by allowing trainees to accumulate operative experience in a zero-risk virtual environment before applying their skills to real patients. Unlike the generic phantom models and cadaveric specimens that currently populate surgical simulation curricula — which provide valuable procedural exposure but cannot replicate the anatomical specificity of individual patient cases — patient-derived digital twins allow the trainee to practice the specific procedure on the specific anatomy they will encounter in the real case, with the specific challenges that the patient’s disease and anatomy present.
The integration of haptic feedback systems — devices that replicate the tactile resistance of biological tissue during virtual instrument interaction — with high-fidelity digital twin models is advancing rapidly, and the near-term prospect is of a training environment in which the trainee can feel the resistance of the virtual aortic wall under suture tension, or the compliance differential between tumor and normal parenchyma during dissection, in a way that approaches the proprioceptive richness of real operative experience. This haptic-augmented digital twin simulation is not a replacement for supervised operative training but a complement to it that allows trainees to enter supervised operative cases with a level of case-specific preparedness that has not previously been achievable.
Implementation Challenges: From Research to Routine Clinical Practice
The translation of AI-generated surgical digital twins from academic demonstration to routine clinical workflow faces several interrelated challenges that are important to acknowledge alongside the technology’s potential. The computational infrastructure required to generate high-fidelity digital twins — the GPU-accelerated processing of segmentation pipelines, the finite element solvers for tissue mechanics, the computational fluid dynamics engines for hemodynamic simulation — demands computing resources that are not currently available within the clinical information technology environments of most hospitals, even in high-resource settings.
The turnaround time between the acquisition of preoperative imaging and the availability of a validated digital twin model must be compatible with the urgency of surgical scheduling — a requirement that creates tension with the computational complexity of high-fidelity simulation. Cloud-based processing architectures, which allow the computationally intensive elements of digital twin construction to be offloaded to remote servers with appropriate data privacy and security protections, are the most promising solution to this constraint, and several commercial and academic platforms are developing clinical-grade cloud-based digital twin services that target turnaround times of twenty-four to forty-eight hours from imaging acquisition to model availability.
The validation of digital twin predictions against real intraoperative findings — the essential empirical step that establishes the clinical trustworthiness of the model — requires systematic prospective data collection across diverse surgical procedures, patient populations, and operative outcomes. This validation infrastructure is currently fragmented across academic centers with different methodological approaches, different imaging protocols, and different outcome definitions, making it difficult to aggregate evidence into the regulatory-grade validation datasets that health authorities will require before approving digital twin systems for widespread clinical deployment. The development of standardized validation frameworks, agreed upon by surgical societies, imaging professional bodies, and regulatory agencies in collaboration with technology developers, is an urgent priority for the field.
The turnaround time between the acquisition of preoperative imaging and the availability of a validated digital twin model must be compatible with the urgency of surgical scheduling — a requirement that creates tension with the computational complexity of high-fidelity simulation. Cloud-based processing architectures, which allow the computationally intensive elements of digital twin construction to be offloaded to remote servers with appropriate data privacy and security protections, are the most promising solution to this constraint, and several commercial and academic platforms are developing clinical-grade cloud-based digital twin services that target turnaround times of twenty-four to forty-eight hours from imaging acquisition to model availability.
The validation of digital twin predictions against real intraoperative findings — the essential empirical step that establishes the clinical trustworthiness of the model — requires systematic prospective data collection across diverse surgical procedures, patient populations, and operative outcomes. This validation infrastructure is currently fragmented across academic centers with different methodological approaches, different imaging protocols, and different outcome definitions, making it difficult to aggregate evidence into the regulatory-grade validation datasets that health authorities will require before approving digital twin systems for widespread clinical deployment. The development of standardized validation frameworks, agreed upon by surgical societies, imaging professional bodies, and regulatory agencies in collaboration with technology developers, is an urgent priority for the field.
Ethical Dimensions: Consent, Responsibility, and the Limits of Simulation
The introduction of AI-generated digital twins into surgical planning raises ethical questions that deserve explicit engagement. The use of a patient’s imaging data to construct a virtual anatomical replica for computational simulation involves the processing of highly sensitive personal health information, and the governance frameworks for data security, patient consent, secondary use authorization, and model retention must be developed to standards that preserve patient trust and comply with applicable data protection regulations. The question of whether a patient must provide specific informed consent for their imaging data to be used in digital twin construction — or whether this falls within the implied scope of clinical care data use — is a jurisdictional and institutional question that lacks uniform resolution.
The question of clinical responsibility when a digital twin simulation provides guidance that is subsequently followed but proves incorrect — when the model predicts a clear surgical corridor that turns out to be occupied by an anomalous vessel — is a medico-legal frontier that existing liability frameworks are not fully equipped to address. The digital twin is a decision-support tool, not a decision-maker, and the responsibility for operative decisions remains entirely with the surgeon who uses it. But the practical reality of clinical decision-making is that quantitatively expressed predictions — particularly when embedded in visually compelling three-dimensional models — can carry an authority that influences decision-making in ways that exceed the model’s validated accuracy, a phenomenon well documented in the clinical literature on algorithmic decision support.
The solution to these ethical challenges is not to limit the deployment of digital twin technology but to develop it with appropriate epistemic humility — building confidence intervals and uncertainty quantification into every model output, training surgeons to engage with digital twin predictions as probabilistic guidance rather than anatomical ground truth, and establishing clear institutional protocols for the circumstances under which digital twin simulation is indicated, how its outputs should be incorporated into operative planning, and how discrepancies between model predictions and intraoperative findings should be documented and fed back into model improvement.
The question of clinical responsibility when a digital twin simulation provides guidance that is subsequently followed but proves incorrect — when the model predicts a clear surgical corridor that turns out to be occupied by an anomalous vessel — is a medico-legal frontier that existing liability frameworks are not fully equipped to address. The digital twin is a decision-support tool, not a decision-maker, and the responsibility for operative decisions remains entirely with the surgeon who uses it. But the practical reality of clinical decision-making is that quantitatively expressed predictions — particularly when embedded in visually compelling three-dimensional models — can carry an authority that influences decision-making in ways that exceed the model’s validated accuracy, a phenomenon well documented in the clinical literature on algorithmic decision support.
The solution to these ethical challenges is not to limit the deployment of digital twin technology but to develop it with appropriate epistemic humility — building confidence intervals and uncertainty quantification into every model output, training surgeons to engage with digital twin predictions as probabilistic guidance rather than anatomical ground truth, and establishing clear institutional protocols for the circumstances under which digital twin simulation is indicated, how its outputs should be incorporated into operative planning, and how discrepancies between model predictions and intraoperative findings should be documented and fed back into model improvement.
Conclusion: The Operating Room of the Future Has a Rehearsal Space
The operating room in Figure 1 will not change because digital twins exist. The tissue will still need to be divided, the vessels controlled, the structures identified and protected by human hands guided by trained eyes and years of accumulated operative judgment. The physical reality of surgery — its irreversibility, its time pressure, its absolute intolerance for anatomical misidentification — is not altered by any amount of computational preparation. It is, in fact, precisely that unforgiving physical reality that makes the case for digital twin-based rehearsal so compelling.
Every high-stakes performance domain that humans have mastered — aviation, military operations, elite athletics, emergency medicine resuscitation — has recognized that optimal performance under pressure requires deliberate preparation that goes beyond theoretical knowledge to include situation-specific rehearsal. Surgery has long been the outlier: a domain whose technical demands rival any of these fields, but whose preparation has historically relied on general anatomical knowledge, case-type experience, and the individual surgeon’s cognitive modeling capacity. The digital twin changes this, bringing to surgical preparation the same principle of specific, scenario-based rehearsal that underlies expert performance in every other high-stakes domain.
The operating room of the future will have a rehearsal space. It will not be a room with a cadaver or a phantom model — though both remain valuable in their respective roles. It will be a computational environment in which the specific patient’s specific anatomy is available for exploration, manipulation, and rehearsal before the patient is on the table. The surgeon who walks into the real operating room having rehearsed the case in that environment will not merely be better prepared. They will, for their patient, be a fundamentally different and more capable instrument of care.
Every high-stakes performance domain that humans have mastered — aviation, military operations, elite athletics, emergency medicine resuscitation — has recognized that optimal performance under pressure requires deliberate preparation that goes beyond theoretical knowledge to include situation-specific rehearsal. Surgery has long been the outlier: a domain whose technical demands rival any of these fields, but whose preparation has historically relied on general anatomical knowledge, case-type experience, and the individual surgeon’s cognitive modeling capacity. The digital twin changes this, bringing to surgical preparation the same principle of specific, scenario-based rehearsal that underlies expert performance in every other high-stakes domain.
The operating room of the future will have a rehearsal space. It will not be a room with a cadaver or a phantom model — though both remain valuable in their respective roles. It will be a computational environment in which the specific patient’s specific anatomy is available for exploration, manipulation, and rehearsal before the patient is on the table. The surgeon who walks into the real operating room having rehearsed the case in that environment will not merely be better prepared. They will, for their patient, be a fundamentally different and more capable instrument of care.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis