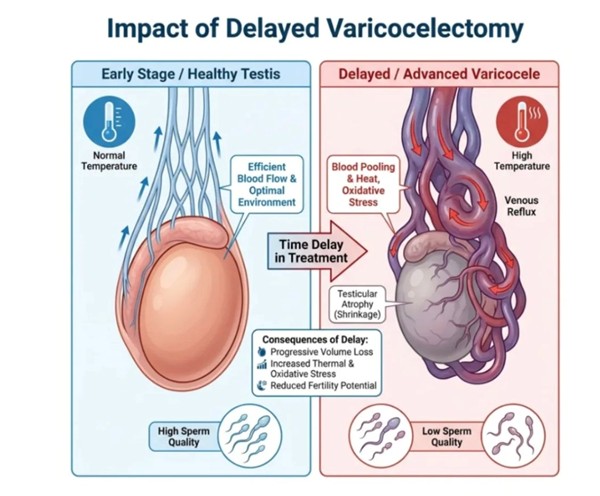
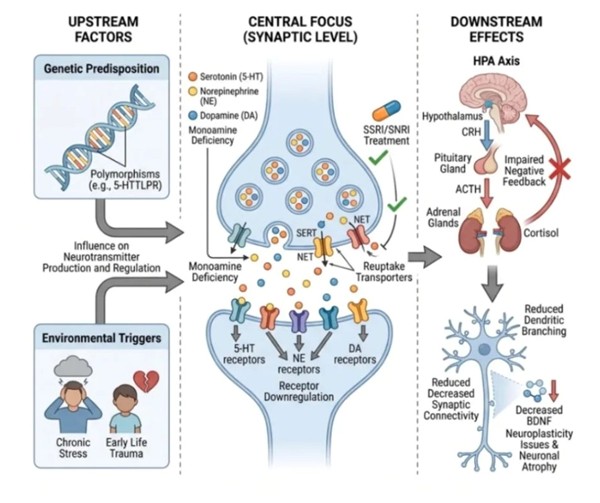
-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
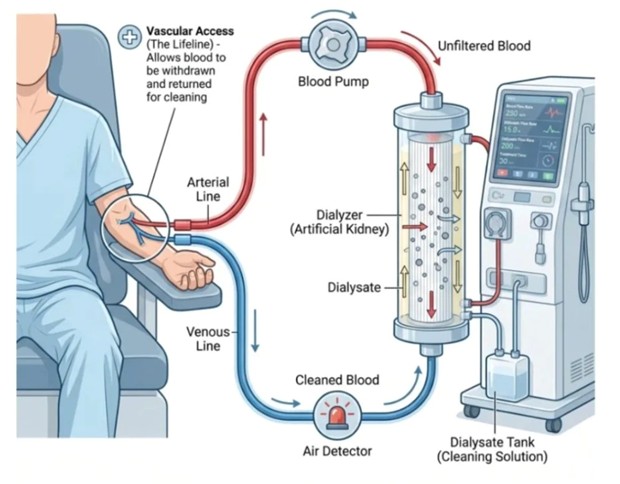
Life Outside the Kidney: Understanding How Hemodialysis Cleans Your Blood
The Lifeline: Establishing Vascular Access for Blood Extraction
Before a single drop of blood can be cleaned, hemodialysis requires something that no medication or dietary change can provide: a reliable, high-flow physical connection to the patient's cardiovascular system. This is the vascular access, and it is rightly described as the lifeline of dialysis therapy. Without it, the entire procedure cannot begin. In patients with end-stage kidney disease, this access is most commonly created surgically in the forearm in one of three forms — an arteriovenous fistula, in which an artery is directly joined to a vein to produce a robust, high-pressure vessel capable of sustaining the rapid blood flows dialysis demands; an arteriovenous graft, which uses a synthetic tube to bridge an artery and vein; or, in acute settings, a central venous catheter placed in a large central vein such as the internal jugular or femoral. Once this access is established and needled, two lines extend from the patient's bloodstream into the external circuit. The arterial line, conventionally marked in red, is the outgoing channel: blood under the pressure of the patient's own cardiovascular system — and assisted by the mechanical blood pump — is continuously drawn out of the body at flow rates typically between 200 and 400 milliliters per minute, beginning its journey through the extracorporeal circuit toward purification.
The Artificial Kidney: Filtration Inside the Dialyzer
The dialyzer is the functional heart of the hemodialysis system — a compact, disposable device that replicates, in a simplified form, the filtration work that the kidneys can no longer perform. Structurally, the dialyzer is a cylinder containing thousands of microscopic hollow fibers, each with walls made from a semipermeable membrane. As the patient's blood flows through the interior of these fibers, a specially formulated cleaning solution called dialysate is pumped simultaneously through the surrounding space of the dialyzer in the opposite direction — a critical design principle known as counter-current flow. This arrangement maximizes the concentration gradient across the semipermeable membrane at every point along the fibers' length, driving the diffusion of waste products, excess electrolytes, and uremic toxins such as urea and creatinine from the blood — where their concentration is high — into the dialysate — where their concentration is intentionally kept low or absent. The dialysate itself is a precisely engineered aqueous solution of bicarbonate, sodium, potassium, calcium, and other electrolytes, mixed in concentrations that guide the correction of not just waste removal but also the electrolyte and acid-base imbalances that accompany renal failure. Simultaneously, excess water that has accumulated in the patient's body between dialysis sessions is removed through ultrafiltration — a pressure-driven process that forces fluid across the membrane in amounts carefully calculated by the dialysis machine to return the patient to their ideal dry weight. The entire filtration process is remarkably efficient: a standard four-hour session processes the patient's entire blood volume many times over, achieving a degree of waste clearance that, while not matching the continuous function of a healthy kidney, is sufficient to sustain life indefinitely.
The Return Circuit: Safety Systems That Guard Every Drop After passing through the dialyzer, the now-purified blood must complete the circuit by returning to the patient's bloodstream — but this return journey is not without its own engineering safeguards
After passing through the dialyzer, the now-purified blood must complete the circuit by returning to the patient's bloodstream — but this return journey is not without its own engineering safeguards. The cleaned blood travels back through the venous line, conventionally marked in blue, which carries it from the dialyzer back toward the vascular access point. Before re-entering the patient, however, it passes through one of the most important safety mechanisms in the entire circuit: the air detector. Any introduction of air bubbles into the venous bloodstream carries the risk of air embolism — a potentially fatal event in which air enters the heart or pulmonary vasculature and disrupts normal cardiac function. The air detector uses ultrasonic technology to continuously monitor the blood flowing through the venous line; at the slightest detection of air or foam, it automatically triggers a clamp on the line and activates an alarm, halting the circuit before any air can reach the patient. This single safety device has transformed what was once a genuine and recurring hazard into an effectively managed risk. The dialysis machine itself adds further layers of protection: continuous real-time monitoring of blood and dialysate flow rates, transmembrane pressure, conductivity of the dialysate solution, blood leak detection, and the patient's vital signs all run simultaneously throughout the session. Together, these interlocking safety systems ensure that hemodialysis — a procedure that sends up to one-third of a patient's blood outside their body at any given moment — can be performed with a safety profile that has made it one of the most routinely performed and life-sustaining medical procedures in the world.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis